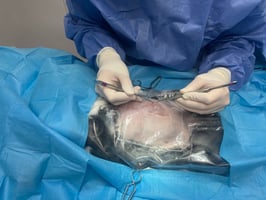
Hyperadrenocorticism has a different pathophysiology and presentation in ferrets compared to dogs and cats. Widely thought of as the most common disease in ferrets (Schoemaker, 2009), hyperadrenocorticism was first described in ferrets in 1987 (Fox et al., 1987). It occurs with excessive reproductive hormone levels, including androsterone, 17 alpha-hydroxyprogesterone and oestradiol (Chitty, 2013).
It is common in surgically neutered ferrets, although it is also reported in entire ferrets. The theoretical pathophysiology is that following neutering and the loss of negative feedback, increased plasma concentrations of gonadotrophins occur and persistently stimulate the adrenal cortex. Following this, adrenal glands become hypertrophied and/or develop tumours (Schoemaker, 2009). Adenomas or adenocarcinomas are the most commonly described tumours of the ferret adrenal gland (Oglesbee, 2011).
Predispositions
One study showed a significant correlation between age at neutering and age of onset of hyperadrenocorticism in ferrets, the median time interval of which was three and a half years (Schoemaker et al., 2000). This study also showed there was no sex predisposition to developing hyperadrenocorticism. It has also been proposed that exposure to increased light levels may increase the chances of developing hyperadrenocorticism (Wagner et al., 2001) as light cycles longer than eight hours stimulate the production of gonadotrophin-releasing hormone (GnRH) and luteinising hormone in ferrets (Jallageas, 1994).
Clinical signs
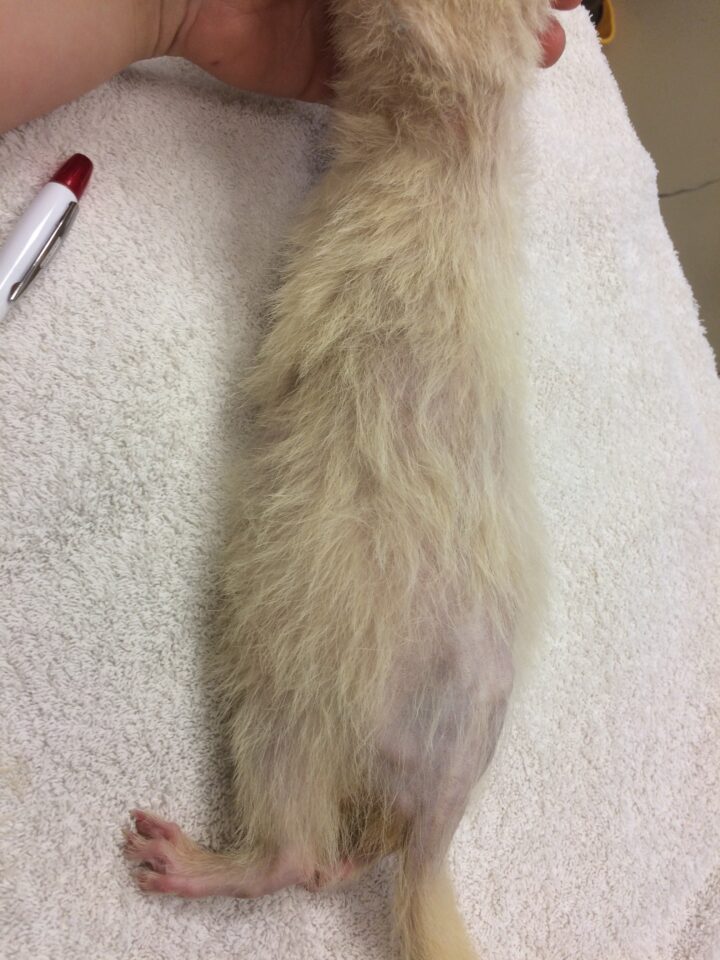
Clinical signs of hyperadrenocorticism in ferrets include thinning of the skin, muscle atrophy, a pot-bellied appearance, sexual aggression and bilaterally symmetrical alopecia with “rat tail” (Figure 1) (Oglesbee, 2011). Alopecia usually begins in spring, at the start of the normal breeding cycle (Schoemaker, 2009). Progressive alopecia of a neutered ferret is considered pathognomonic for the disease (Chitty, 2013).

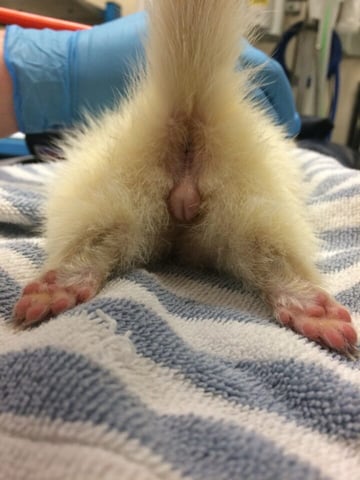
A swollen vulva (Figure 2) or mammary hyperplasia can be seen in neutered females, and stranguria can be observed in males due to prostatic hyperplasia, urogenital cysts or prostatic abscesses (Oglesbee, 2011).
Diagnosis
Diagnosis of hyperadrenocorticism in ferrets is often guided by clinical signs (Schoemaker, 2009).
Unlike canine and feline patients, dexamethasone suppression tests and ACTH stimulation tests are of no diagnostic value in ferrets. Ferret “adrenal panels” are offered by several commercial laboratories in the UK to test androstenedione, oestradiol and 17 alpha-hydroxyprogesterone. Elevation of any of these hormones is considered diagnostic for hyperadrenocorticism (Schoemaker, 2009). However, hormone concentrations in intact females are identical to ferrets with hyperadrenocorticism, so results must be interpreted with caution even in neutered females, who may have ovarian remnant syndrome (Quesenberry and Rosenthal, 2003).
Urine corticoid:creatinine ratio is elevated in ferrets with hyperadrenocorticism but is also elevated in ferrets with ovarian remnant syndrome, so is of limited use in female ferrets (Schoemaker, 2013).
Imaging
Ultrasound is a useful tool to visualise the adrenal glands and assess if one or both are enlarged (Oglesbee, 2011). Guidelines for identifying both adrenal glands have been described, which allows veterinarians to assess the size of each (Kuijten et al., 2007). Normal adrenal glands usually measure 2 to 3.7mm wide and 4 to 8mm long (Oglesbee, 2011). Adrenal glands are abnormal if they have an enlarged cranial or caudal pole (over 3.9mm), have a rounded appearance, show increased echogenicity and/or contain mineralisation (Figure 3) (Kuijten et al., 2007).
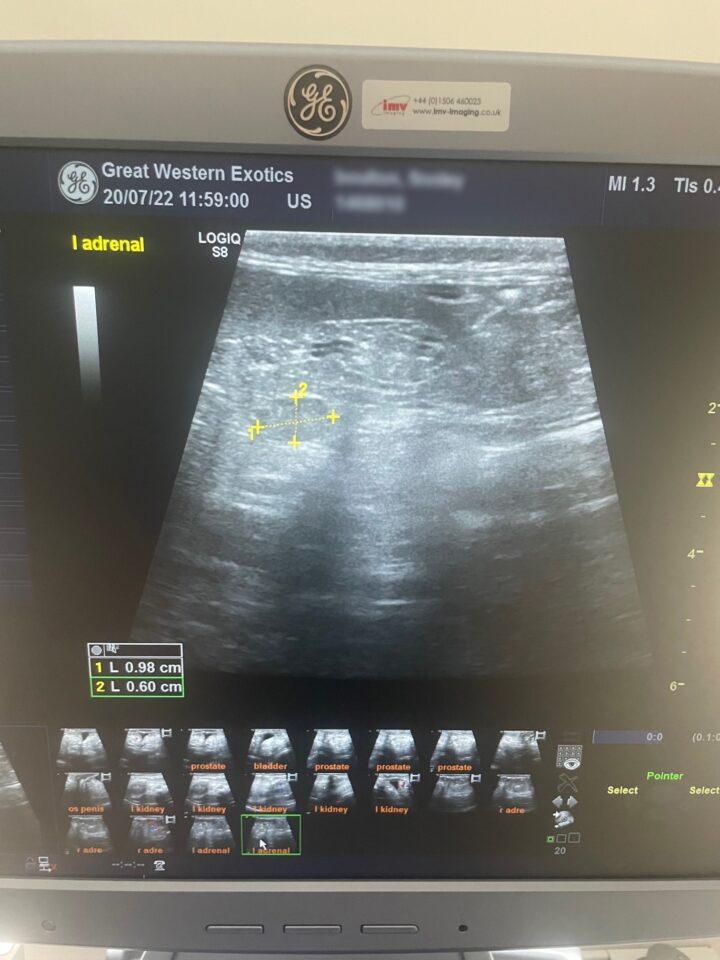
Computed tomography (CT) can also be used as an imaging modality to assess adrenal gland size and other comorbidities. However, the benefits of ultrasound over CT include the lack of requirement for sedation or general anaesthesia (as most ferrets will sit happily for an abdominal ultrasound when distracted with liquid food or “ferret oil”) and that ultrasound is very accessible in general clinical practice.
Treatment
Once diagnosed, two main treatments are available to treat hyperadrenocorticism in ferrets: surgical or non-surgical. The choice of treatment can depend on several factors, including the client’s preference and experience, the patient’s age, anaesthetic suitability, treatment costs and the veterinary surgeon’s experience.
Surgery
Surgical adrenalectomy used to be the treatment of choice, with the affected adrenal gland identified via diagnostic imaging. The surgery for adrenalectomy on either side has been well described (Beeber, 2011); however, anatomical differences between the two glands make the removal of the right adrenal gland more complicated.
The right adrenal gland lies close to the caudal vena cava, and during resection it may be necessary to leave a portion of the gland attached to the vena cava
The right adrenal gland lies close to the caudal vena cava, and during resection it may be necessary to leave a portion of the gland attached to the vena cava (Schoemaker, 2009). This makes full resolution of adrenal disease questionable, and inappropriate partial ligation of the caudal vena cava can result in hypertension distal to the surgical site, which can cause acute renal failure (Schoemaker, 2009).
Surgery is, however, usually associated with good outcomes. A retrospective study of 130 ferrets that underwent surgical adrenalectomy for hyperadrenocorticism showed one-year survival rates of 98 percent and two-year survival rates of 88 percent (Swiderski et al., 2008).
Medical management
Alternatively, medical management is less invasive but relies on clients to be observant and closely monitor their ferrets for signs of recurrence (Chen et al., 2014).
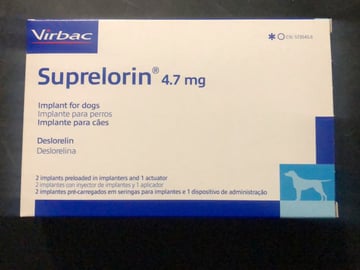
GnRH implants are commonly used to manage hyperadrenocorticism in ferrets and offer a potentially safer and more effective alternative to surgery (Lennox and Wagner, 2012). A single 4.7mg GnRH-agonist deslorelin implant (Figure 4) was shown to significantly decrease reproductive hormones associated with hyperadrenocorticism and decrease or resolve clinical signs of hyperadrenocorticism, such as vulval swelling, pruritus and aggression (Wagner et al., 2009). However, the same study also showed the mean duration of action was 17.6 ± 5.0 months, with a range of 8 to 30 months. This means that ferrets will very likely need repeat deslorelin implants throughout their lifetime to continue suppressing reproductive hormones.
Deslorelin implants have been reported to have minimal side effects. However, case reports do exist of pregnancy following deslorelin implantation (Cecchetto et al., 2019) and implant-associated soft tissue sarcoma (Hollwarth et al., 2021).
Lupron depot injections have also been used in the USA to control hyperadrenocorticism, with a duration of action of one to three months (Schoemaker, 2009).
GnRH-agonist use may seem counter-intuitive as hyperadrenocorticism is caused by an excess of reproductive hormones, but their success is related to the method of hormone release. The release of gonadotrophins in response to GnRH relies on a pulsatile release of GnRH, so when a steady release occurs, the pulsatile release does not occur, resulting in a single release of gonadotrophins followed by no further release (Schoemaker, 2009). It is also for this reason that clinicians should warn owners that they may see a transient increase in sexual behaviours before improvement after placement of a deslorelin implant.
GnRH-agonist use may seem counter-intuitive as hyperadrenocorticism is caused by an excess of reproductive hormones, but their success is related to the method of hormone release
GnRH-agonist use may seem counter-intuitive as hyperadrenocorticism is caused by an excess of reproductive hormones, but their success is related to the method of hormone release
Melatonin has been proposed for the medical management of hyperadrenocorticism, as ferrets are long-day breeders (Jallageas, 1994). Release of GnRH is increased by increasing day length, so the administration of melatonin is theorised to simulate shorter day length, therefore reducing the release of GnRH and inhibiting the release of luteinising hormone and follicle-stimulating hormone (Chen et al., 2014).
Options for non-breeding ferrets
With the increased risk of ferrets developing adrenal disease following surgical neutering, owners often face issues when keeping groups of mixed-sex ferrets. If breeding is not the goal, several options remain available to owners.
Vasectomising hobs (male ferrets) has the dual benefit of providing non-fertile males and the ability to use these hobs to bring jills (female ferrets) out of season in spring. This is important as jills are induced ovulators, and lack of ovulation can result in potentially fatal hyperoestrogenism (Schoemaker, 2009). However, this option will not reduce sexual aggression between ferrets in a group and can lead to injuries between hobs and in jills being mated by hobs.
The preventative use of deslorelin GnRH-agonist implants is now commonly recommended over surgical neutering. A 9.4mg Suprelorin (deslorelin) implant is licensed in the UK for chemical neutering of male ferrets, but 4.7mg and 9.4mg Suprelorin implants can be used off-licence in female ferrets. These implants have been shown to be successful in both sexes not only to reduce sexual-related behaviours but also to prevent the onset of hyperadrenocorticism by downregulating hormonal control pathways, preventing adrenal hypertrophy from occurring in the first place (Chitty, 2013).
In ferrets that are already neutered, for example those being adopted from animal charities where neutering prior to adoption is the protocol, deslorelin implants can also be used to downregulate hormonal pathways and prevent hyperadrenocorticism successfully (Chitty, 2013).
Summary
Ferret hyperadrenocorticism is significantly different to hyperadrenocorticism presenting in canine and feline patients. Surgically neutered ferrets have a higher risk of developing disease, but it has been identified in surgically neutered and non-neutered ferrets. Several treatment options are available; however, implantation with GnRH-agonist implants has been shown to be safe, effective and minimally invasive.
References (click to expand)
Ashton Hollwarth, BSc, BVMS, CertAVP (Zoo Med), ANZCVS (Avian Medicine & Surgery), DipECZM (Avian), MRCVS, studied in Western Australia before moving to the UK after graduating. Following a short time in general practice, she completed her residency in avian medicine and surgery at Great Western Exotics. Ashton currently works exclusively in avian and exotics, and her clinical interests include avian and exotic anaesthesia, parrot behaviour and small mammal surgery. Ashton gained her Certificate of Advanced Veterinary Practice in Zoological Medicine in 2020, became an Advanced Practitioner in Zoological Medicine in 2021 and gained membership by examination to the Australian and New Zealand College of Veterinary Scientists’ Avian Chapter in 2022. She became a diplomat of the European College of Zoological Medicine as a specialist in avian medicine and surgery in 2024.